 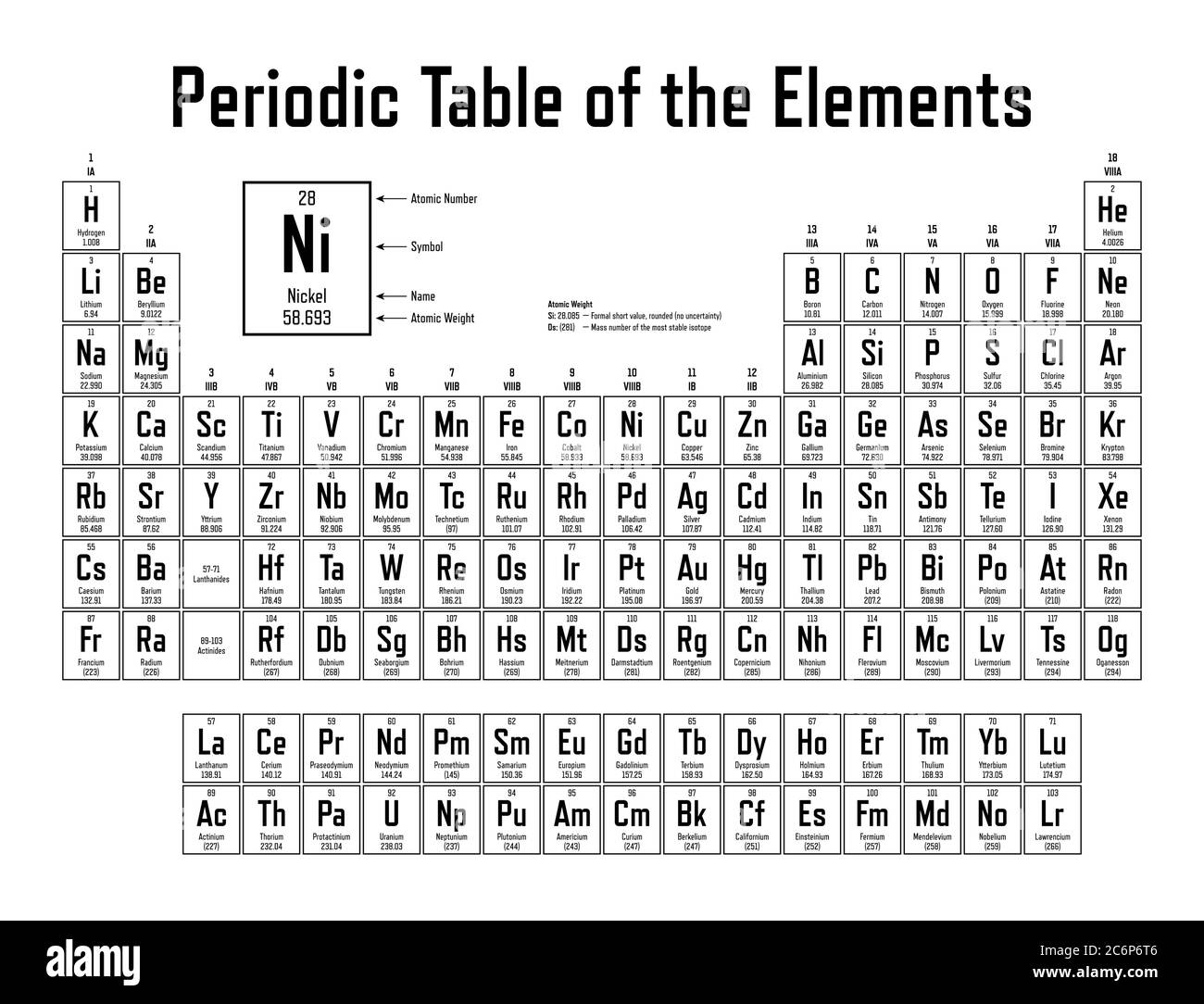
Mendeleev’s version of the periodic table left the biggest impact on the scientific community, both at the time it was produced and thereafter. However, there was an untimely delay in the publication of his most elaborate periodic table, and, perhaps more importantly, Meyer-unlike Mendeleev-hesitated to make predictions about unknown elements. 
The closest precursor to Mendeleev’s table in both chronological and philosophical terms was developed by Julius Lothar Meyer, a German chemist, in 1864. By the 1860s a number of scientists had moved beyond the triad concept to produce some very respectable periodic systems. The elements in these groupings displayed an important numerical relationship to each other: the equivalent weight (an early substitute for atomic weight) of the middle element had the approximate mean of the values of the two flanking elements. The observation that certain types of elements prefer to combine with certain other types prompted early chemists to classify the elements in tables of chemical affinities. Mendeleev was hardly the first to arrive at a periodic system. It marks the 150th anniversary of the publication by the Russian chemist Dmitry Mendeleev (1834–1907) of his Periodic Table and celebrates the significance and impact of this outstandingly successful chart of the atomic building blocks of matter. The year of 2019 was designated by the United Nations as the International Year of the Periodic Table of Chemical Elements. This periodic table chart lists elements by name in alphabetical order including the element symbol and atomic number for quick and simple reference. Periodic Table with Element Names periodic table in alphabetical order by symbol 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
